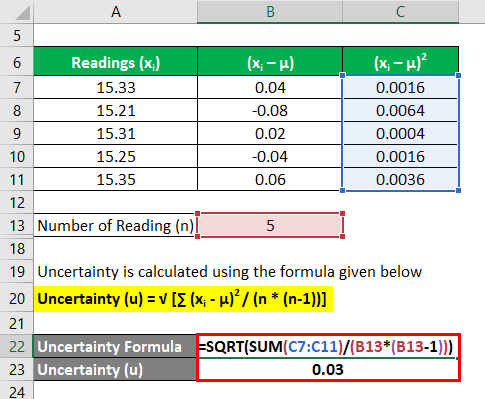
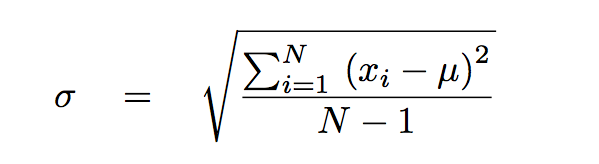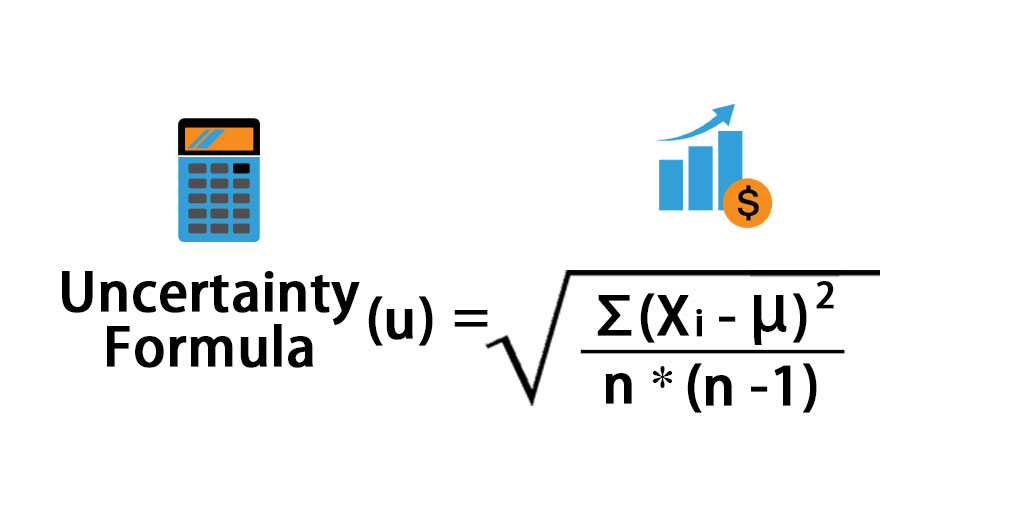The ± 1 second is called the absolute uncertainty Every measurement has an uncertainty or error. e.g. time = 5 seconds ± 1 second There are three main. - ppt download

1.2 Uncertainties and errors Random/systematic uncertainties Absolute/fractional uncertainties Propagating uncertainties Uncertainty in gradients and intercepts. - ppt download

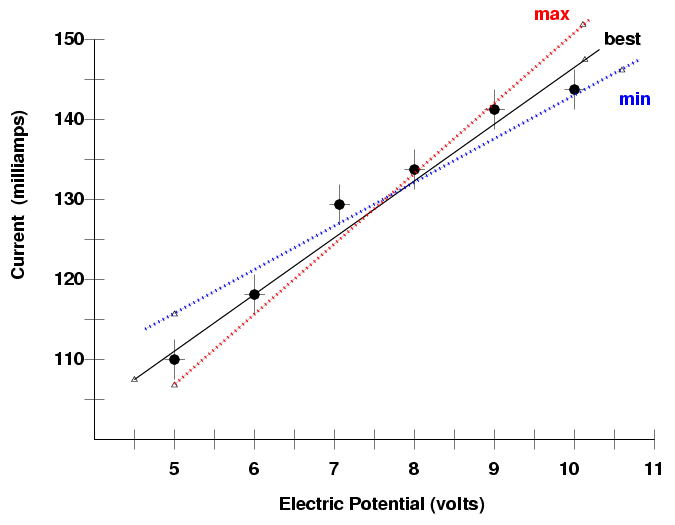
Determining Uncertainties from Graphs (1.2.3) | AQA A Level Physics Revision Notes 2017 | Save My Exams




![11.1 State uncertainties as absolute and percentage uncertainties [SL IB Chemistry] - YouTube 11.1 State uncertainties as absolute and percentage uncertainties [SL IB Chemistry] - YouTube](https://i.ytimg.com/vi/wrnCMUP8V1o/maxresdefault.jpg)