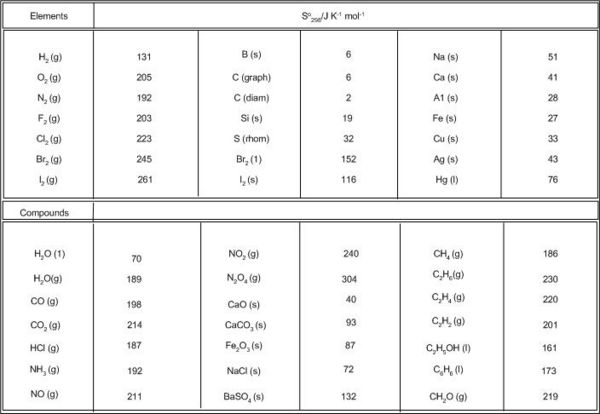
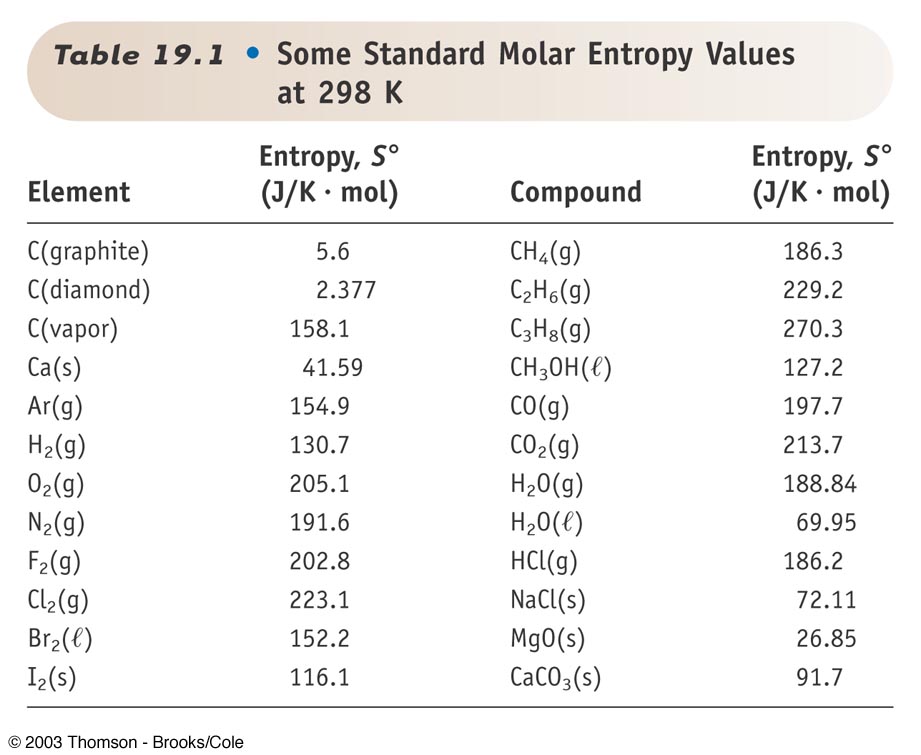
SOLVED: * Required 1. Calculate IS? at 25*C for the reduction of Pbo(s), 2Pbo(s) + C(s) > 2Pb(s) + CO2(g) given these absolute entropies S' (J/K-mol) Entropy values Pbo(s)= 69.45 C(s) =

OneClass: Calculate the standard entropy change for the reaction P_4(g) + 5O_2(g) rightarrow P_4 O_10...
![The Determination of Absolute Values of Entropies of Hydration [ΔSabs0(H+)h] and Aquation [ΔSabs0(H+)aq] and The Thermodynamics of Proton in Solutions The Determination of Absolute Values of Entropies of Hydration [ΔSabs0(H+)h] and Aquation [ΔSabs0(H+)aq] and The Thermodynamics of Proton in Solutions](https://www.degruyter.com/document/doi/10.1515/zpch-2016-0867/asset/graphic/j_zpch-2016-0867_fig_001.jpg)
The Determination of Absolute Values of Entropies of Hydration [ΔSabs0(H+)h] and Aquation [ΔSabs0(H+)aq] and The Thermodynamics of Proton in Solutions

Why is negative the absolute entropy, enthalpy and internal energy in solids on the sublimation curve (steam properties)? | ResearchGate

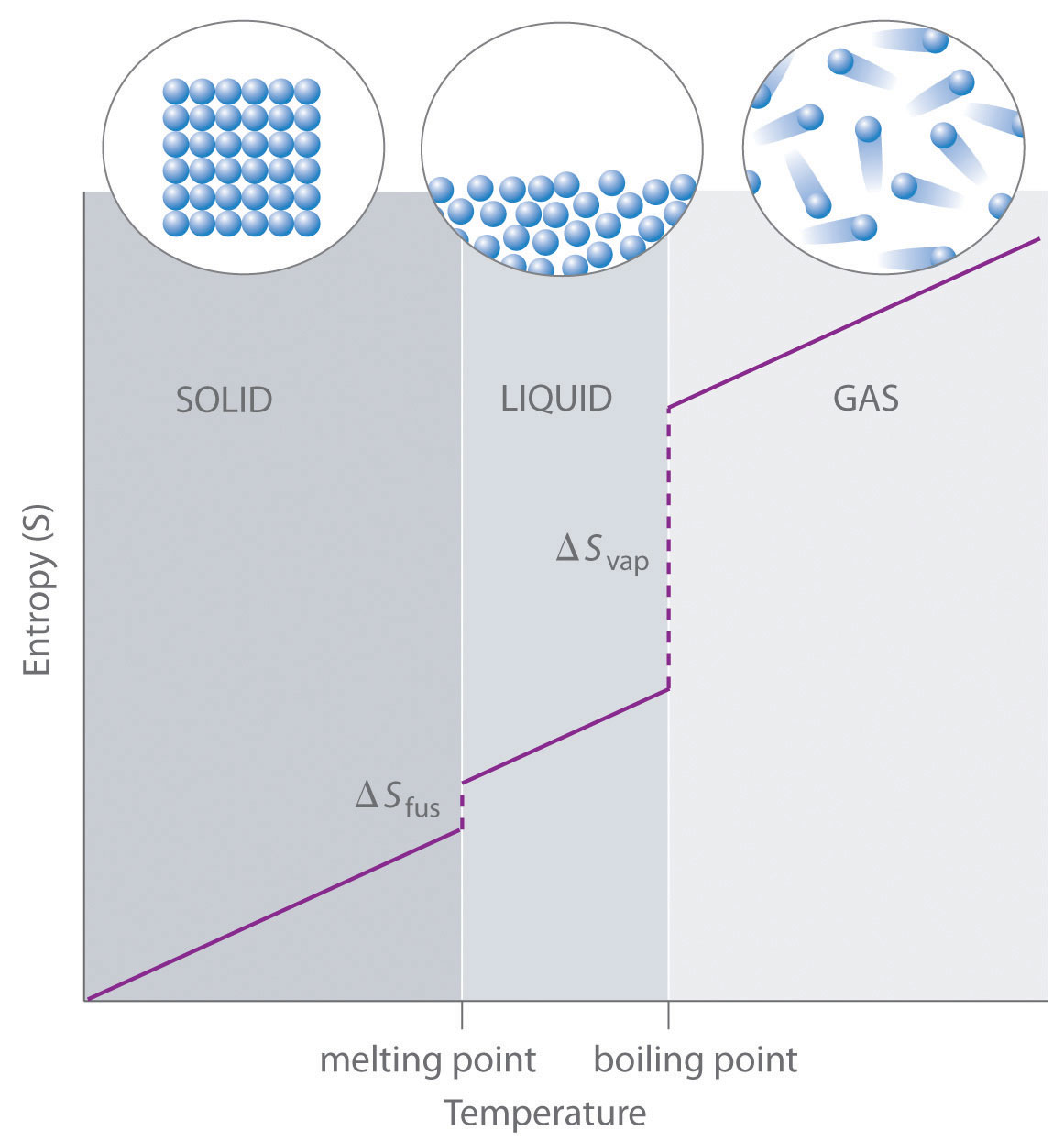
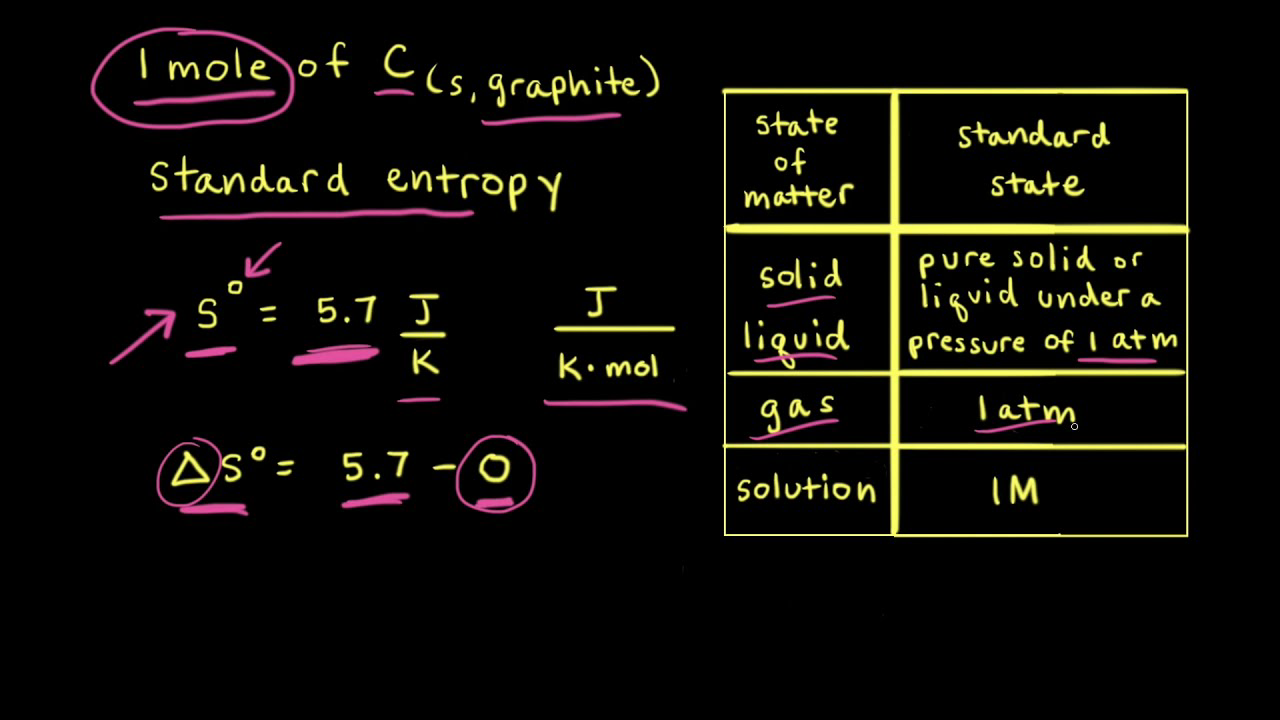
Determination of Absolute Entropy Values for Solid, Liquid & Gases with third law of thermodynamics. - YouTube

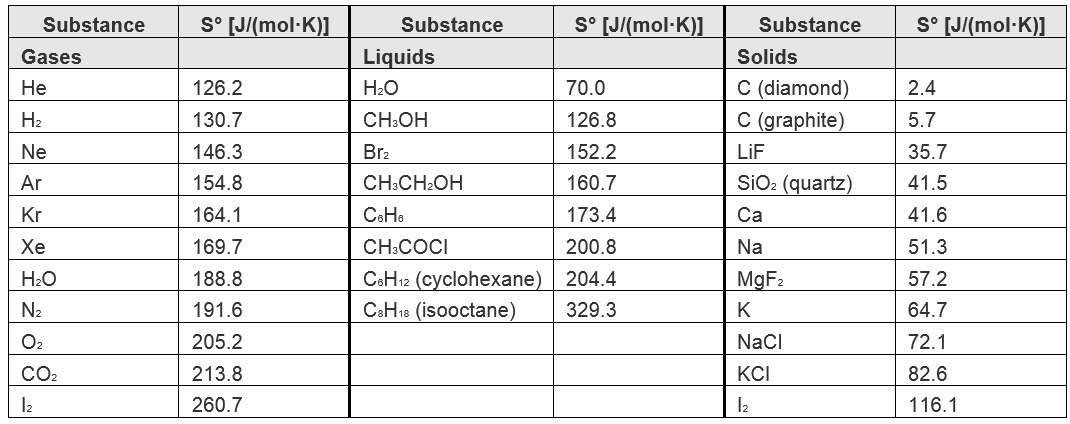
Standard Absolute Entropy, , Values from Volume or Density. 1. Inorganic Materials | Inorganic Chemistry
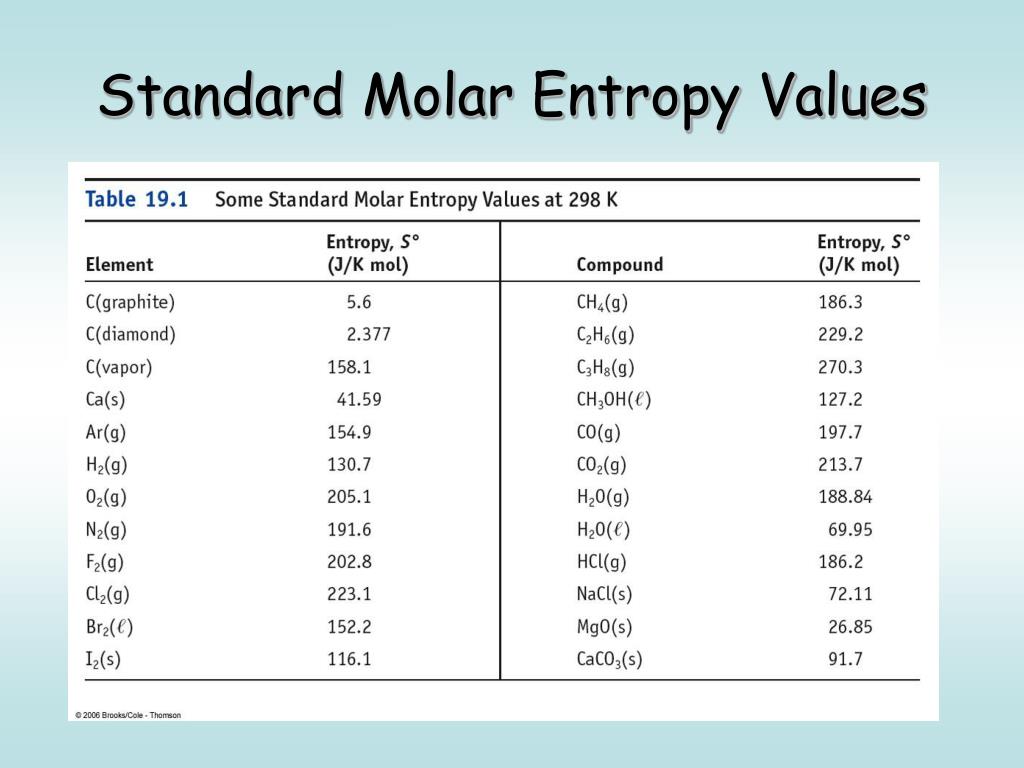
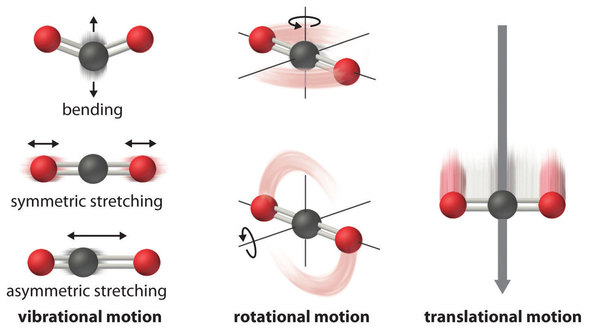
PPT - Chapter 19 – Principles of Reactivity: Entropy and Free Energy PowerPoint Presentation - ID:1293047