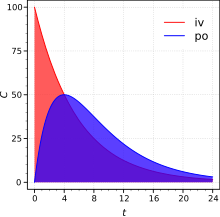
![The Absolute Bioavailability, Absorption, Distribution, Metabolism, and Excretion of BI 425809 Administered as an Oral Dose or an Oral Dose with an Intravenous Microtracer Dose of [14C]-BI 425809 in Healthy Males | SpringerLink The Absolute Bioavailability, Absorption, Distribution, Metabolism, and Excretion of BI 425809 Administered as an Oral Dose or an Oral Dose with an Intravenous Microtracer Dose of [14C]-BI 425809 in Healthy Males | SpringerLink](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs40261-021-01111-9/MediaObjects/40261_2021_1111_Fig1_HTML.png)
The Absolute Bioavailability, Absorption, Distribution, Metabolism, and Excretion of BI 425809 Administered as an Oral Dose or an Oral Dose with an Intravenous Microtracer Dose of [14C]-BI 425809 in Healthy Males | SpringerLink

Assessment of Oral Bioavailability and Biotransformation of Emulsified Nobiletin Using In Vitro and In Vivo Models | Journal of Agricultural and Food Chemistry

Absolute bioavailability, dose proportionality, and tissue distribution of rotundic acid in rats based on validated LC-QqQ-MS/MS method - ScienceDirect
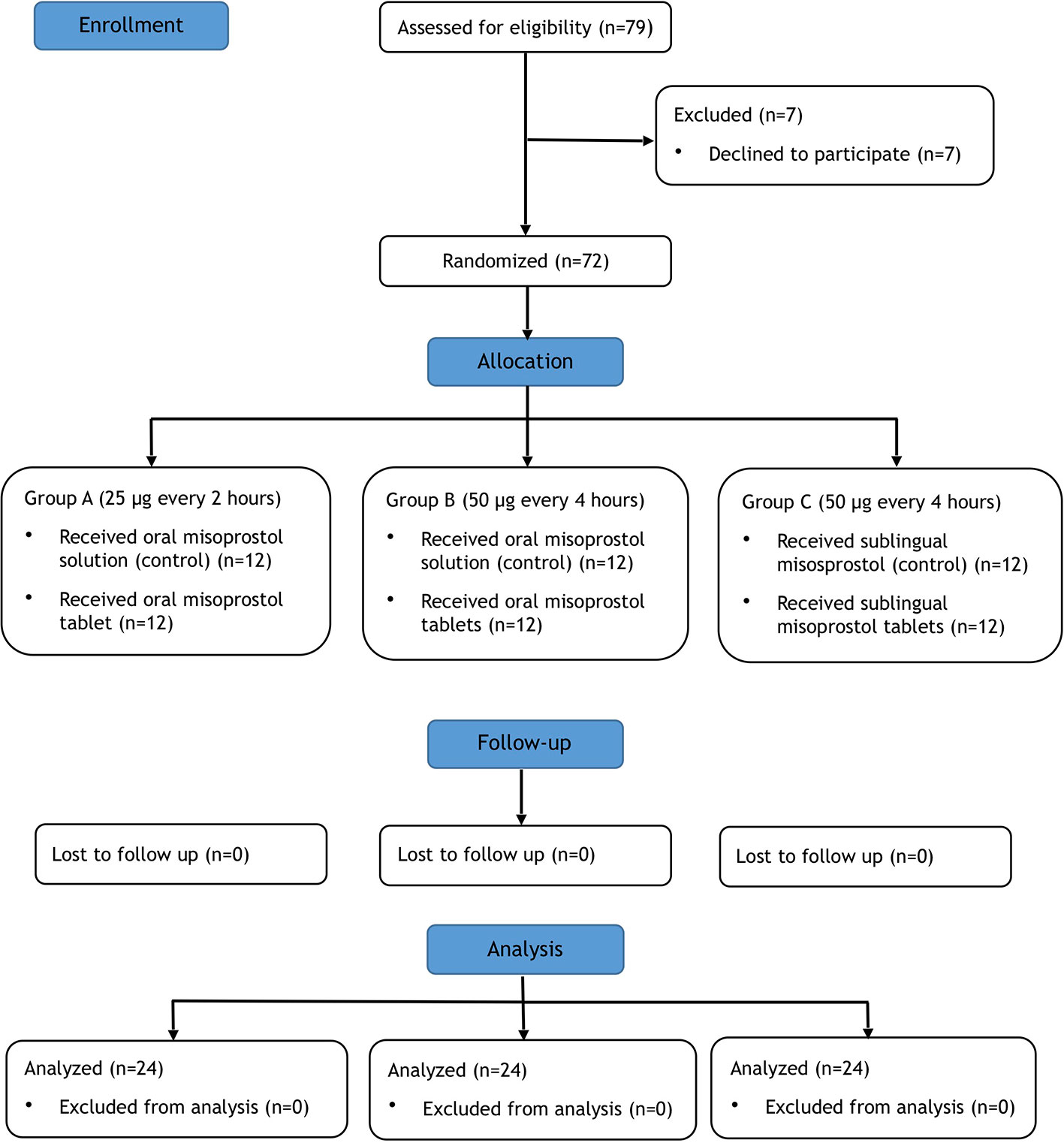
Frontiers | A Relative Bioavailability Study of Two Misoprostol Formulations Following a Single Oral or Sublingual Administration

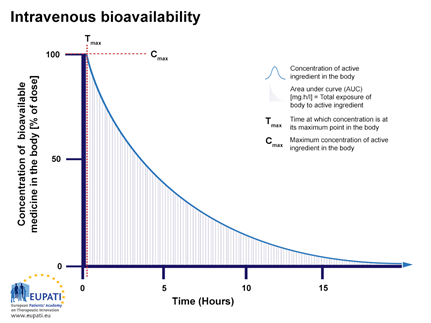
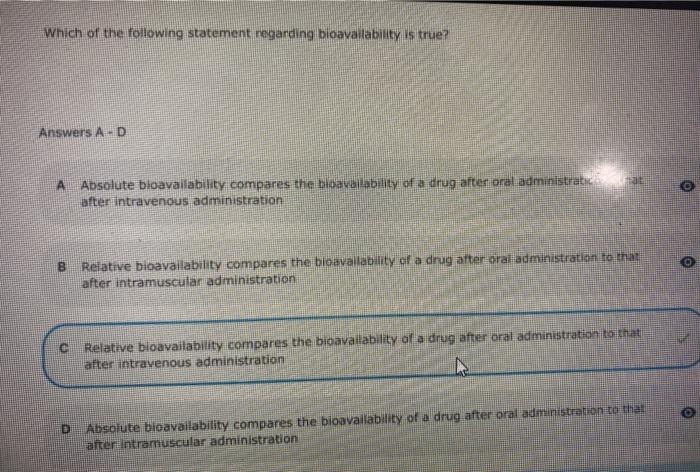
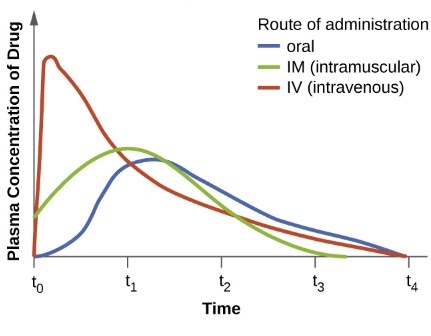
Bioavailability , absolute bioavalability, relative bioavailability, Purpose of bioavailability, Methods of assesing bioavailability
![Overcoming bioanalytical challenges in an Onglyza® intravenous [14C]microdose absolute bioavailability study with accelerator MS | Bioanalysis Overcoming bioanalytical challenges in an Onglyza® intravenous [14C]microdose absolute bioavailability study with accelerator MS | Bioanalysis](https://www.future-science.com/cms/10.4155/bio.12.171/asset/images/medium/figure2.gif)
Overcoming bioanalytical challenges in an Onglyza® intravenous [14C]microdose absolute bioavailability study with accelerator MS | Bioanalysis
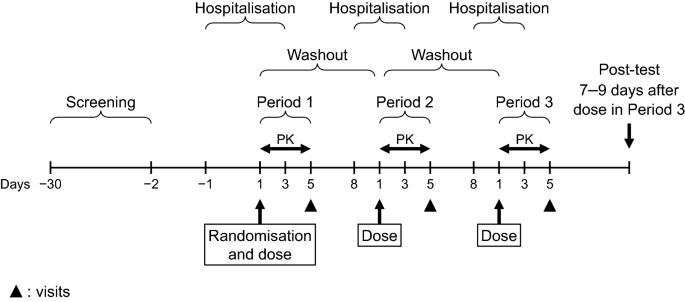
Absolute Bioavailability of Esaxerenone and Food Effects on its Pharmacokinetics After a Single Oral Dose in Healthy Japanese Subjects: An Open-Label Crossover Study | SpringerLink

Bioavailability- 1 Bioavailability Metrological aspects Assessment of parameters Update: july /211/ ppt download

Sensitivity-based analytical approaches to support human absolute bioavailability studies | Bioanalysis
ABSOLUTE AND RELATIVE BIOAVAILABILITY STUDY FOR THE NEWLY DEVELOPED NASAL NANOEMULSION IN SITU GEL OF ONDANSETRON HCl IN COMPARI